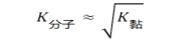耐火材料结构中的敏感因素–透气度
在压力差下,使气体渗过多孔物体的性质称作透气性。在黏滞流动制度(牛顿)的气体中,出口压力接近大气压,并且用柱形形式气孔测定气孔率大于5%的耐火材料的透气度,按泊肃叶公式计算k=Qh/A△p。在国际制系统中透气度系数用m3表示。它相当于横断面为1m2,长1m的试样,气体体积为1m3,黏度为1Pa·s,压力差为 1Pa时,通过1s穿过时,取得的透气度。实际单位使用um2。计算公式为: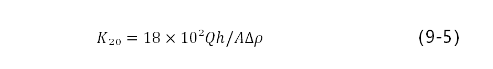
式中,k20为温度为20℃的透气度系数μm2,Q为20℃时通过试样的空气流量cm3/s;h为试样高度(进入的深度);A为试样断面面积,cm3△p为试样中进口和出口之间的压力差,Pa。
以下计算取得系数
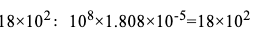
式中108为重新计算的系数,平方微米中的平方厘米:1.808×10-5为20℃时空气的动态黏度,压力差△p大于2kPa时,按公式(9-5)得出的结果乘2(p+p)/(2p+Δp)
式中,p为入口空气的压力,Pa。
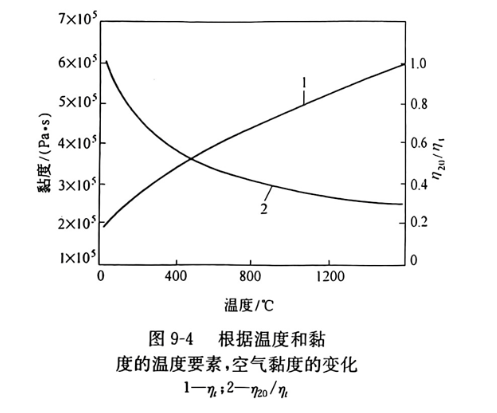
最大的透气度相当于气孔尺寸20~100um。这个等级的气孔范围几乎包括所有透气的气孔。气孔尺寸愈大,透气度也愈大。制品的结构对透气度非常敏感。如某些结构的变化,导致开口气孔率2倍的变化,透气性变化 100倍。高温时制品的透气度(Kt)按下列公式:

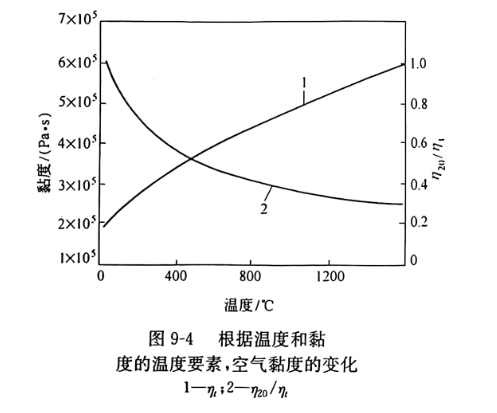
式中,K20为按公式(9-5)的透气度系数;n20/nt为室温时的气体黏度对某温度时黏度的比率(图9-4)。所有耐火材料(与类型无关)高温时的透气度明显下降,800℃与20℃时的透气度比较大的约为一半。
按空气透气性测定透气度,相当于氮,氧,二氧化碳和高炉气体的透气度,因为这些气体的黏度大体相同。
工业耐火制品的透气度系数值处在以下范围:黏土质(0.2~1.0)μm2,硅质的(0.1~1.2)μm2,镁质的((0.6~1.2)μm2,铬镁质的(0.8~2.5)μm2。致密制品(气孔率<5%)的透气度在气体分子(努森)流动制度中测定。气体分子流动制度取决于压力和通道尺寸。为了整个通道均匀,尺寸小于103μm通道气体在分子制度中流动,气体压力应该是 1.33Pa和更低些。分子流动时,制品结构要给予气流最小的阻力,因此结构参数一样时,
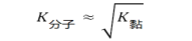
Permeability – Sensitive factor in refractory structure
Under pressure differences, the property of allowing gas to permeate porous objects is called permeability. In the gas of viscous flow system (Newton), the outlet pressure is close to atmospheric pressure, and the permeability of refractories with porosity greater than 5% is measured by cylindrical form porosity, calculated k=Qh/A△p according to Poiseuille formula. In the international system, the air permeability coefficient is expressed as m3. It is equivalent to a specimen with a cross-section of 1m2, a length of 1m, a gas volume of 1m3, a viscosity of 1Pa·s, and a pressure difference of 1Pa, and the permeability obtained when passing through 1s. The actual unit uses um2. The calculation formula is:
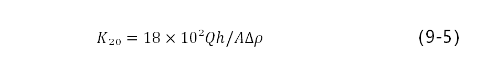
Where, k20 is the permeability coefficient μm2 at a temperature of 20℃, and Q is the air flow rate cm3/s through the sample at 20℃. h is the sample height (depth of entry); A is the section area of the sample, cm3△p is the pressure difference between the inlet and outlet of the sample, Pa.
The following calculation obtains the coefficients
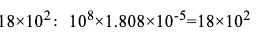
Where 108 is the recalculated coefficient, the square centimeter in the square micron :1.808×10-5 is the dynamic viscosity of the air at 20℃, and when the pressure difference △p is greater than 2kPa, multiply the result obtained by the formula (9-5) by 2(p+p)/(2p+Δp).
Where, p is the pressure of the inlet air, Pa.
The maximum permeability is equivalent to the porosity size of 20~100um. The range of pores in this class includes almost all air vents. The larger the pore size, the greater the permeability. The structure of the product is very sensitive to air permeability. Such as some structural changes, resulting in a two-fold change in the porosity of the opening and a 100-fold change in air permeability. The air permeability (Kt) of the product at high temperature according to the following formula:

Where, K20 is the permeability coefficient according to the formula (9-5); n20/nt is the ratio of gas viscosity at room temperature to viscosity at a certain temperature (Figure 9-4). The air permeability of all refractories (regardless of type) at high temperature is significantly reduced, and the air permeability is about half of that at 800℃ and 20℃.
Air permeability is measured by air permeability, which is equivalent to the air permeability of nitrogen, oxygen, carbon dioxide and blast furnace gases, because these gases have roughly the same viscosity.
The air permeability coefficient of industrial refractory products is in the following range: clay (0.2~1.0) μm2, silicon (0.1~1.2) μm2, magnesium (0.6~1.2) μm2, chrome-magnesium (0.8~2.5) μm2. The air permeability of dense products (porosity <5%) is determined in the gas molecular (Knudsen) flow system. The flow regime of gas molecules depends on the pressure and channel size. In order to be uniform throughout the channel, the size of the channel gas less than 103μm flow in the molecular system, the gas pressure should be 1.33Pa or lower. When the molecule flows, the structure of the product should give the minimum resistance to the airflow, so when the structural parameters are the same,