耐火材料对钢中非金属夹杂物的影响
在耐火材料的应用中,不同物质相互作用下会产生不同属性,按钢中非金属夹杂物的来源分为3组:
(1)内生的——金属本身,当它脱氧,结晶化和冷却时,由于进行各种物理化学过程,形成内部起源;
(2)外成的——由耐火材料和渣构成外部起源,当液体金属流放出和浇注时,机械诱导而可能凝固在钢中。最危险的是液体金属诱导耐火材料小质点,因为大质点容易浮起;
(3)内外生的——在外成上分泌出,当时内生夹杂物。用目力按标准等级表的级数(图 11-18)评价钢的非金属夹杂物的污染性。在增大100~125 倍的显微镜下观察抛光的显微磨片,与标准等级表比较,轧钢试样总合起来。用这个方法主要确定外成夹杂物,这种夹杂物在所有夹杂物的份额中相对不大。
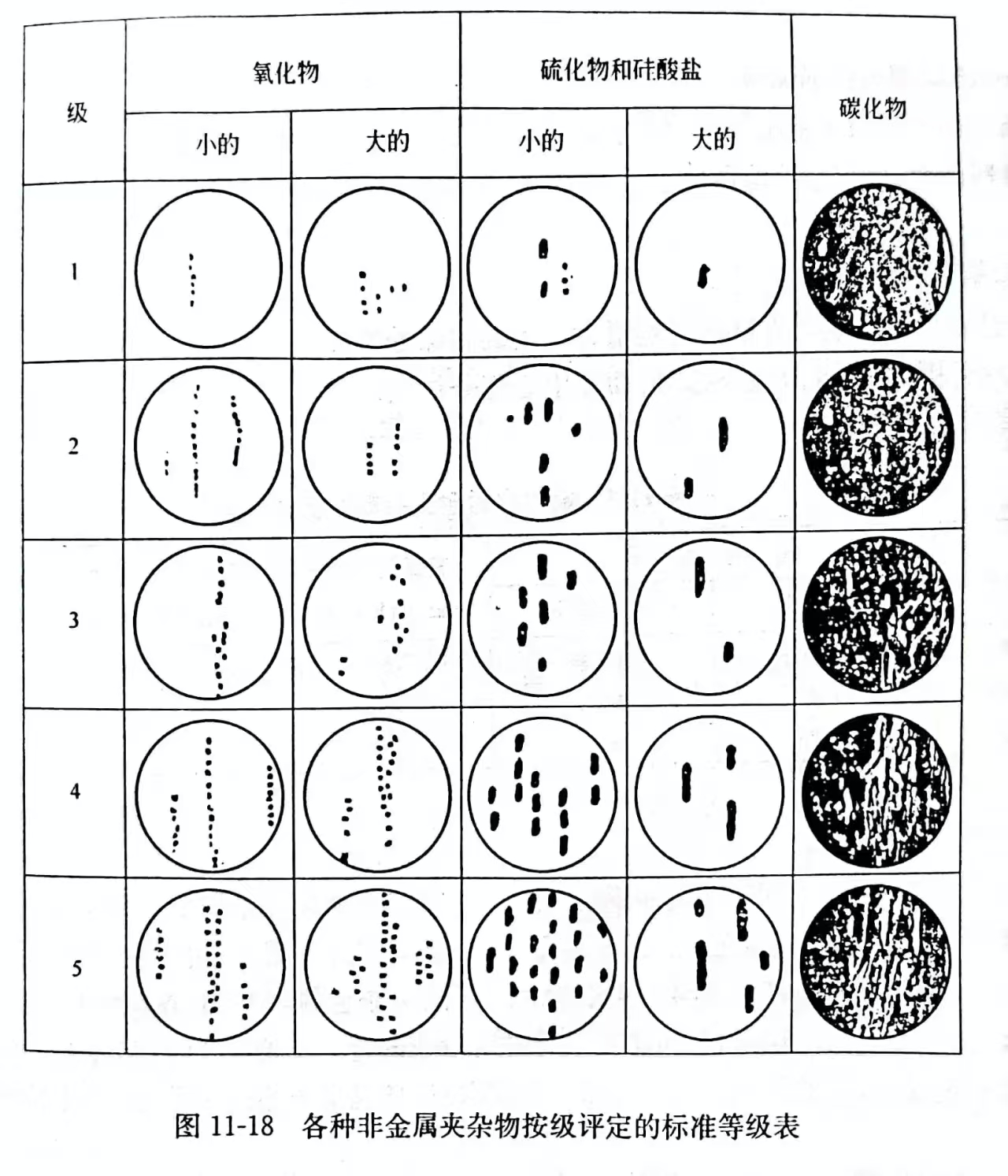
正常冶金过程,外成夹杂物不超过夹杂物总量的 5%~10%。利用放射性同位素(示踪原子)多次研究这个令人信服的指标。所有夹杂物的质量分数一般为0.01%~0.02%。
密度最小的非金属夹杂物能浮起。浮起一般遵循已知的司托克规则。可实际上夹杂物尺寸不大时,黏附力有很大的作用。润湿性的程度愈小(金属——夹杂物的相间能很大),抑制夹杂物与金属接触的力就愈小,而它愈易与金属分开,并且浮起。作为夹杂物的例子,液体铁润湿不好的,可能遭受氧化铝夹杂物(σ金-Al2O3≈1J/m2);作为夹杂物的例子,液体铁润湿好的(所以与它不好分开),可能遭受硅酸铁夹杂物(σ金-杂≈0.4J/m2)。尺寸为1~2μm的夹杂物上升得很慢,由于气流对流,可能长时间在液体金属体积中转移,而最终结果仍旧在金属中。通过熔池散发的气泡(金属沸腾,惰性气体吹炼熔池等)与夹杂物拥有的相间能比夹杂物与金属之间的相间能小,即(σ杂-气<σ金-杂),所以夹杂物对气泡黏着,并与它消失在渣中。
关于夹杂物从金属排除速度的影响:
(1)夹杂物的尺寸,它的成分(熔化温度)和密度;
(2)对夹杂物增大的能力;
(3)金属-夹杂物和渣-夹杂物边界上的相间能;
(4)熔池的搅拌强度;
(5)金属的物理性能。
一、出钢槽内衬的影响出钢槽内衬被大量损坏:1t钢为2~6kg。放出一炉由 10min延长到20min。这个时间冲刷的耐火材料碎片,由于金属层厚度不大而浮起。因此由出钢槽内衬耐火材料污染钢的可能性很小。
二、钢包内衬的影响已经表现出钢包耐火材料消耗很大,大的和深处的夹杂物浮起。此外,钢包中钢发生回转移动。钢在外围部分变冷,往下移动,而在中心部分往上。同时,某些部分的非金属夹杂物仍旧在钢中,见表11-5。
|
表11-5钢包内衬耐火材料对钢的污染 |
|||||
|
钢包砖 |
砖的性质 |
紧要侧的损毁/mm·(h·热)-1 |
按氧化物夹杂物、钢的平均级数 |
夹杂物对钢污染,砖参与的质量分数/% |
|
|
Al2O3/% |
气孔率/% |
||||
|
黏土砖 |
36.4 |
19.2 |
11 |
1.82 |
4.6 |
|
高岭石砖 |
44.41 |
10.5 |
4.4 |
1.62 |
2.5 |
|
高铝砖 |
77.73 |
5.6 |
3.6 |
1.56 |
1.5 |
三、流钢砖的影响统计资料表明:用流钢砖法浇注钢锭比上注的钢锭非金属夹杂物少。多孔流钢砖甚至吸收钢的铝脱氧形成的内生非金属夹杂物的某些部分;可又有另一观点,由于流钢砖的热稳定性低,因此崩裂,是流钢砖的过错产生非金属夹杂物。为了减少非金属夹杂物,在浇注硅钢时,建议采用半硅材料,浇注锰含量高的钢时,采用游离氧化硅含量小的砖。无论怎样,浇钢砖应该是热稳定性的,只有玻璃相量最小,它的结构才合乎愿望,即流钢砖应该是低温烧成多孔的,或是不烧的。
四、钢水炉外精炼的影响虽然在钢水炉外精炼过程中耐火材料剧烈损坏,它参与钢中非金属夹杂物的形成,在这里是最低限度的,因为炉外精炼过程本身的发展方向,就是降低所有类型的非金属夹杂物。耐火材料溶解在金属中时,耐火材料元素转为离子或原子形式。这些元素在金属中,与气体相互作用形成非金属夹杂物,于是形成氮化物,氧化物,硫化物等。这样一来,耐火材料在钢中的任何溶解都形成非金属夹杂物。
Effect of refractory materials on non-metallic inclusions in steel
In the application of refractory materials, different substances will produce different properties under the interaction, according to the source of non-metallic inclusions in steel is divided into 3 groups:
(1) Endogenous – the metal itself, when it deoxidizes, crystallizes and cools, forms an internal origin due to various physicochemical processes;
(2) Exogenetic – composed of refractory materials and slag of external origin, when the liquid metal is exiled and poured, mechanically induced and may solidify in the steel. The most dangerous is the liquid metal induced refractory small particles, because large particles are easy to float;
(3) Endogenous – exogenous secretion on exogenous, when endogenous inclusions. Visually evaluate the pollution of non-metallic inclusions in steel according to the level of the standard grade table (Figure 11-18). The polished microgrinding plate is observed under a microscope with an increase of 100 to 125 times, and the rolled steel sample is compared with the standard grade table. This method is used primarily to identify exogenous inclusions, which are relatively small in the share of all inclusions.
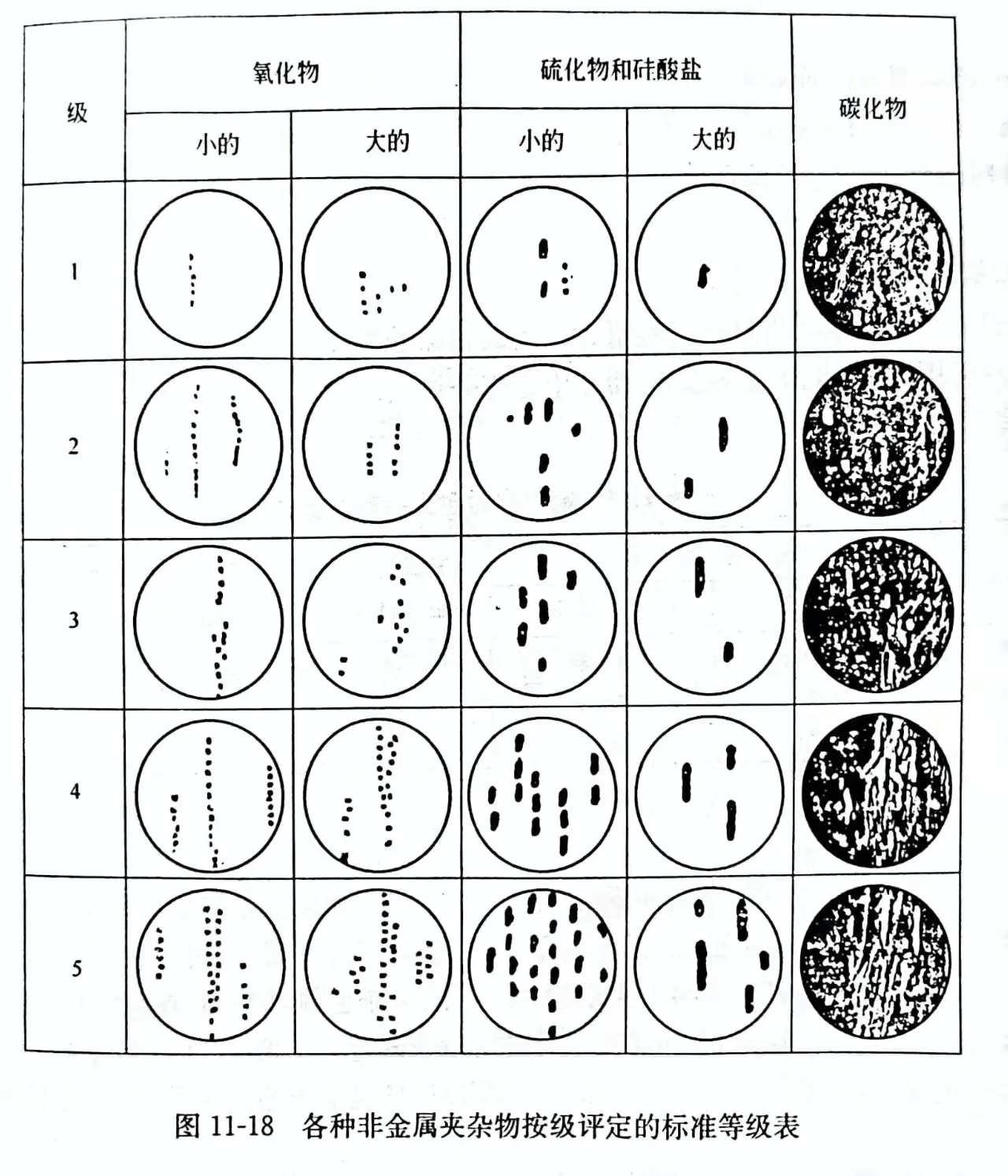
In normal metallurgical process, the external inclusion does not exceed 5% to 10% of the total inclusion. This compelling indicator has been studied many times using radioisotopes (tracer atoms). The mass fraction of all inclusions is generally 0.01%~0.02%.
The least dense nonmetallic inclusions can float. Buoyancy generally follows what is known as Stocker’s rule. But in fact, when the inclusion size is not large, the adhesion force has a great effect. The smaller the degree of wettability (the large interphase energy of the metal-inclusions), the smaller the force that restrains the contact between the inclusions and the metal, and the easier it is to separate from the metal and float. As an example of inclusion, liquid iron is not well wetted and may suffer from alumina inclusion (σ-al2o3 ≈1J/m2); As an example of an inclusion, liquid iron is well wetted (so is not well separated from it) and may suffer from an inclusion of iron silicate (σ-au ≈0.4J/m2). Inclusions with a size of 1-2 μm rise very slowly and, due to airflow convection, may be transferred in the liquid metal volume for a long time, while the end result remains in the metal. The bubbles emitted through the molten pool (metal boiling, inert gas blowing molten pool, etc.) and the inclusion have a smaller phase energy than the phase energy between the inclusion and the metal, that is, (σ-gas < σ-gold – impurity), so the inclusion sticks to the bubble and disappears with it in the slag.
On the effect of exclusion rate of inclusions from metal:
(1) the size of the inclusion, its composition (melting temperature) and density;
(2) the ability to increase inclusions;
(3) interphase energy at metal-inclusion and slag-inclusion boundaries;
(4) stirring intensity of the molten pool;
(5) Physical properties of metals.
First, the impact of the steel groove lining The steel groove lining is a lot of damage: 1t steel is 2~6kg. The discharge of a furnace is extended from 10min to 20min. The fragments of refractory material washed out at this time float due to the small thickness of the metal layer. Therefore, the possibility of contaminating the steel by the refractory lining of the steel groove is very small.
Second, the impact of ladle lining has shown that ladle refractory material consumption is large, and large and deep inclusions float. In addition, the steel in the ladle rotates. The steel cools in the outer part and moves down, while it goes up in the center part. At the same time, some parts of non-metallic inclusions remain in the steel, as shown in Table 11-5.
|
Table 11-5 Steel pollution caused by refractory materials in ladle lining |
|||||
|
Ladle brick |
Properties of brick |
Critical side damage /mm·(h· heat)-1 |
According to the average grade of oxide inclusions and steel |
Inclusion of steel contamination, brick participation in the mass fraction /% |
|
|
Al2O3/% |
Porosity/% |
||||
|
Clay brick |
36.4 |
19.2 |
11 |
1.82 |
4.6 |
|
Kaolinite brick |
44.41 |
10.5 |
4.4 |
1.62 |
2.5 |
|
High alumina brick |
77.73 |
5.6 |
3.6 |
1.56 |
1.5 |
The statistical data show that the ingot cast by the method of flowing steel bricks has less non-metallic inclusions than the ingot cast. Porous flow steel bricks even absorb some parts of endogenous nonmetallic inclusions formed by aluminum deoxidation of steel; But there is another point of view, because of the low thermal stability of the flow steel brick, so the crack is the fault of the flow steel brick to produce non-metallic inclusions. In order to reduce non-metallic inclusions, it is recommended to use semi-silicon materials when pouring silicon steel, and use bricks with small free silicon oxide content when pouring steel with high manganese content. In any case, the cast steel brick should be heat stable, only the glass phasor is minimal, its structure is in line with the desire, that is, the flow steel brick should be low temperature fired into porous, or not fired.
Although the refractory material is severely damaged during the refining process of the molten steel furnace, it is involved in the formation of non-metallic inclusions in the steel, which is the minimum here, because the development direction of the refining process itself is to reduce all types of non-metallic inclusions. When the refractory is dissolved in the metal, the refractory elements change into ionic or atomic form. In metals, these elements interact with gases to form nonmetallic inclusions, thus forming nitrides, oxides, sulfides, etc. In this way, any dissolution of the refractory in the steel forms non-metallic inclusions.



